PROPERTIES
Activated carbon is the generic term used to describe a family of carbonaceous adsorbents with a highly crystalline form and extensively developed internal pore structure. A wide variety of activated carbon products is available exhibiting markedly different characteristics depending upon the raw material and activation technique used in their production. In selecting an activated carbon, it is important to have a clear understanding of both the adsorptive and physical characteristics of the material in order to optimise the performance capabilities.
PHYSICAL PROPERTIES OF CARBON
Particle size distribution:
The finer the particle size of an activated carbon, the better the access to the surface area and the faster the rate of adsorption kinetics. In vapour phase systems this needs to be considered against pressure drop, which will affect energy cost.
Careful consideration of particle size distribution can provide significant operating benefits.
The important properties of activated carbon relevant to specific applications are considered further in the text.
Pores:
Activated carbon has a large volume of very small pores which creates a large surface area. Typical activated carbons have surface areas from 600 to 1,200 m2/g with some reported as high as 3,000 m2/g. These internal pores are classified based on size as either micropores (10 to 1,000 A) or macropores (greater than 1,000 A). Adsorption occurs primarily in the micropores with the macropores acting as conduits.
 |
 |
Iodine and Molasses Number:
The iodine number provides an indication of the amount of small pores in carbon. It is defined as the milligrams of iodine (I2) that are adsorbed per gram of carbon when the equilibrium concentration of the bulk saturation (Ceq) is 0.02 N. It is also correlated with the surface area in pores with diameters less than 10 Angstroms. Because iodine is a small molecule, it provides an indication of a particular carbon's capacity to adsorb smaller molecules.
The molasses number, in contrast, represents the amount of large pores in carbon. It is calculated from the ratio of the optical densities of thee filtrate of a molasses solution treated with a standard activated carbon and one treated with the activated carbon in question. The molasses number can be correlated with the surface area in pores with diameters greater than 10 Angstroms. Because the color pigments in molasses are large molecules, this information provides an indication of a particular carbon's capacity to adsorb larger molecules.
For compounds which do not have published isotherms, the iodine and molasses numbers may be used as a relative guideline (not an adsorption rate) for measuring the capacity of a carbon for larger adsorbate molecules. Both the iodine number and the molasses number decrease with time as adsorption occurs.
Apparent Density:
The particle density (dry) increases with time as adsorption fills the particle's pores. An associated increase in apparent density, defined as the mass per unit volume of a container occupied by non stratified dry carbon, will result.
Corrosion:
Granular activated carbon is electrically conductive and can set up a galvanic cell which then corrodes carbon steel. The materials of construction can be selected exclusively on the basis of the corrosiveness of the fluid to be treated and the conditions used for regeneration.
Hardness:
An important factor in system design, filter life and product handling. There are large differences in the hardness of activated carbons, depending on the raw material and activity level.
Bulk Density:
Should be carefully considered when filling fixed volumes as it can have considerable commercial implications.
The backwashed and drained density will show a lower value due to the water film between the particles of activated carbon.
ADSORPTIVE CHARACTERISTICS
Surface area (BET N2)
Measurement, using nitrogen (N2), of the extent of the pore surface developed within the matrix of the activated carbon. Used as a primary indicator of the activity level, based on the principle that the greater the surface area, the higher the number of adsorptive sites available.
Carbon tetrachloride activity
Measurement of the porosity of an activated carbon by the adsorption of saturated carbon tetrachloride vapour.
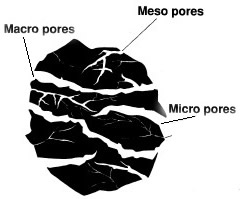
Pore size distribution |
 |
PRIVATE
Micropores r < 1nm
Mesopores r 1-25nm
Macropores r > 25nm
The macropores are used as the entrance to the activated carbon, the mesopores for transportation and the micropores for adsorption.
| Up |